Digital Health Driven Therapy Systems
ACMIT is putting main emphasis on the development of new technology for minimally and micro-invasive procedures with increased consideration of surgical data aspects with the slogan “Digital Health Driven Therapy Systems“.
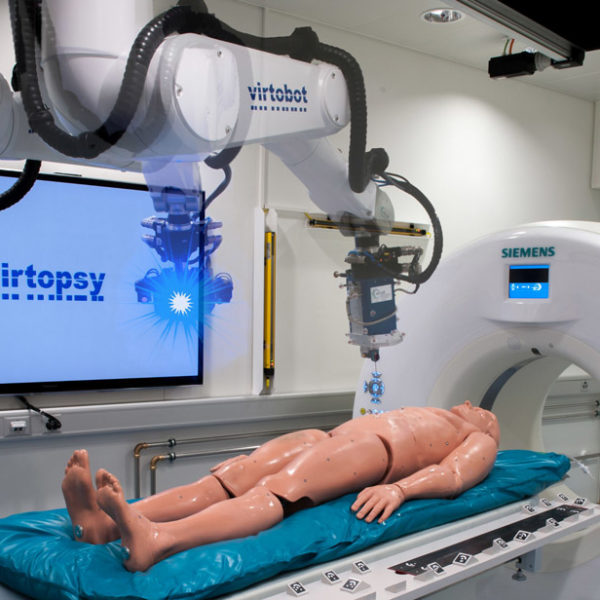
From technical and “procedural” viewpoint, research and development in ACMIT is covering the entire chain of activities – starting from advanced planning of the therapy, over the delivery of therapy by smart tools (e.g. with in-situ monitoring of therapy, advanced control of the therapy process) and guidance of such tools (e.g. by means of medical robotic systems or navigation technology), up to aspects related to the operator site (e.g. work-flow support) and even beyond (e.g. establishing of a surgical knowledge base for a more global advancement of surgical procedures). To complete the chain of translational research and development, work in ACMIT also includes evaluation of such new technology in realistic clinical setups which also addresses the increased requirements from the EU Medical Device Regulation (MDR). ACMIT research and development activities are structured into three research areas. The research scope has been mapped onto a “Sense – Plan – Act” structure.
Your Contact Person

Gernot Kronreif
Prof. h.c. Dipl.-Ing. Dr.techn.
Chief Scientific Officer
+43 2622 22859 0gernot.kronreif@acmit.at
Autonomy in Surgery
This research area is dealing with tools and methods for delivery of treatment, i.e. for the transfer of planned action into action – also including methods for adaptation of planned actions based on situation awareness. In the deliberative control scheme of a therapy process, this area is covering the “acting” phase.
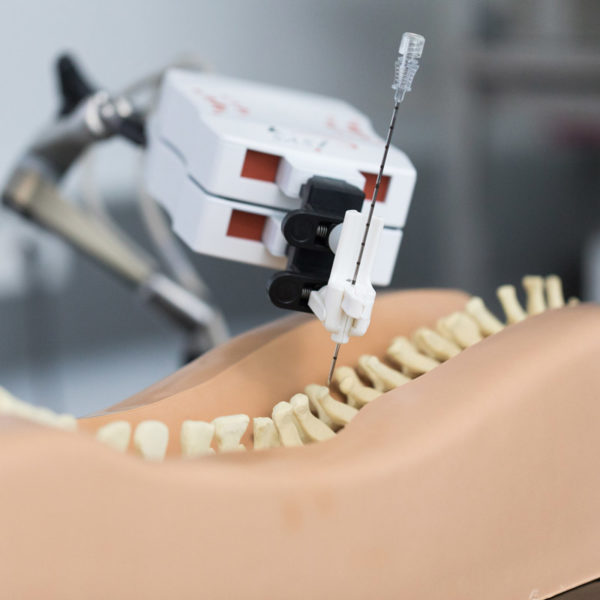
Research tasks of topic “Smart Tools and Instruments” address some of the key points for improvement of state of the art MIS procedures. Based on the existing concept for steerable stylets and trocars, tools will be developed for concrete applications and tested clinically. Tool design activities also will be related to integration of the sensors developed in research area Sensor Systems for Diagnosis and Therapy into surgical tools for real-time in vivo therapy monitoring. Finally, investigation of new surgical access systems will complete the range of activities regarding “smart tools”. Furthermore, new application setups for our existing robot technology will be investigated as well as concepts for Cognitive Surgical Systems. For the latter topic, especially aspects of autonomous task execution as well as collaborative human/robot setups will be analysed in terms of concrete clinical use cases.
Tasks of topic “Information-based Therapy” aim to investigate concepts for data-based treatment. New methods for surgical navigation will be developed, including efficient and accurate registration, soft tissue navigation, and navigation based on real-time imaging. Furthermore, we address treatment optimization based on sensor data in a local context, as well as globally by investigation of methods related to “Surgical IoT” and “Surgical Data Science”.
Your Contact Person

Gernot Kronreif
Prof. h.c. Dipl.-Ing. Dr.techn.
Autonomy in Surgery, Sensor Systems
+43 2622 22859 0gernot.kronreif@acmit.at
Surgical Data for Optimized Therapy
"Surgical Data Science" (SDS) is an emerging field and a major contribution to the current trend towards “Digitalized Medicine” and “Surgery 4.0”. SDS comprises the structured acquisition of relevant data across the entire chain of diagnostic and therapeutic actions and the use of the resulting data pools in order to optimize therapeutic procedures.
The COMET Module “Surgical Data for Optimized Therapy” (SD-OpT) by ACMIT is representing an important extension of ACMIT’s research areas, such as sensor-integrated tools, medical IoT or patient-specific planning tools, which provide an ideal base for “Surgical Data Science” (SDS).
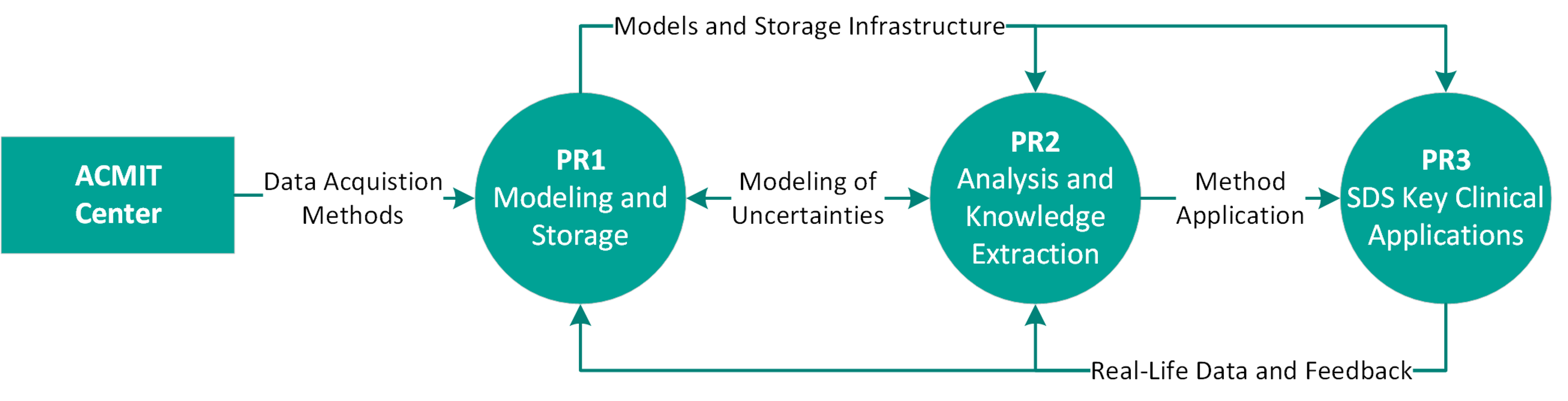
SD-OpT investigates methods of manual and automatic knowledge extraction from data streams and analysis of application-specific data in order to identify relevant correlations to optimize diagnosis and treatment. SD-OpT demonstrates the immense potential of SDS by realization of representative and meaningful evaluation cases. The results from SD-OpT will be integrated by ACMIT into advanced “Digital Health driven Therapy Systems”.
The R&D activities are structured into three projects:
- PR1: Information Modeling, Storage and Distribution
- PR2: Data Analysis and Knowledge Extraction
- PR3: Evaluation of SDS in key clinical applications
SD-OpT establishes a grand coalition of leading institutes in key areas of SDS - a close cooperation of international high-profile partners from science and industry.
Scientific Partners:
- Delft University of Technology, Department of BioMechanical Engineering (Delft, NL)
- German Cancer Research Center DKFZ (Heidelberg, DE)
- Innovation Center for Computer-Assisted Surgery ICCAS (Leipzig, DE)
- INSERM/MediCIS, University of Rennes (Rennes, FR)
- Tokyo Women’s Medical University, ABMES (Tokyo, JP)
- University of Innsbruck, Department of Computer Science (Innsbruck, AT)
Key Researchers:
- Dr. Pierre Jannin, MediCIS/INSERM - University of Rennes
- Dr. Thomas Neumuth, ICCAS- University of Leipzig
- Dr. Keno März, DKFZ Heidelberg
Company Partners:
- EBG MedAustron GmbH (Wiener Neustadt, AT)
- Erbe Elektromedizin GmbH (Tübingen, DE)
- Evaluation Software Development GmbH (Innsbruck, AT)
- Richard Wolf GmbH (Knittlingen, DE)
- BHS Technologies GmbH (Innsbruck, AT)
Your Contact Person

Tamás Haidegger
Prof. Dipl.-Ing. Dr.techn.
Therapy Planning and Workflow Support, Surgical Data Science
+43 2622 22859 0tamas.haidegger@acmit.at
Sensor Systems for Diagnosis and Therapy
This research area is mainly dealing with the development of novel sensor technology for therapy monitoring and thus covers the “sensing” phase in the deliberative control scheme of a therapy process.
This research area is addressing the topics “Advanced Sensor Systems” and “Improving Quality of Vision”.
“Advanced Sensor Systems” essentially aim to provide intra-operative surgeon feedback by system/instrument integrated sensor components. Giving surgeons a real-time feedback about the actually treated tissue can significantly improve the intervention or directly provide diagnostic results. Planned activities include in particular the development of miniaturized sensor components for integration in instruments for minimal invasive surgery, for example for needle based sensorized tools. Together with sensor data acquisition and signal analysis these topics constitute the main research activities. Another part of activities is related to sensorized systems and tools which are not directly involved in surgical interventions. These tasks activities for point of care applications which are settled in the fields of prevention and home care monitoring. Their goal is to provide patient individual data for diagnosis and therapy related decision-making.
Based on our optic design know-how in combination with simulations tools, the topic “Improving Quality of Vision“ deals with development of optical elements under the umbrella of the term “Quality of Vision”. Main focus of activities is in ophthalmology, especially including optic design concepts for intraocular lenses (IOLs) in combination with methods and tools for assessment of the subjective vision quality and patient satisfaction. Multifocal as well as extended depth of focus IOLs not only allow to satisfy individual preferences in cataract surgery, but also entail the need for increased patient guidance, which has to be supported by appropriate tools during the clinical workflow practice. Therefore, lens design for capsular bag IOLs as well as supplementary lens platforms belong to the main research tasks. Besides these activities in ophthalmology, we work on optical setups and components which are not directly dedicated to a sensory functionality, such as improvement of the surgeon’s limited view in endoscopic applications.
Your Contact Person

Gernot Kronreif
Prof. h.c. Dipl.-Ing. Dr.techn.
Autonomy in Surgery, Sensor Systems
+43 2622 22859 0gernot.kronreif@acmit.at
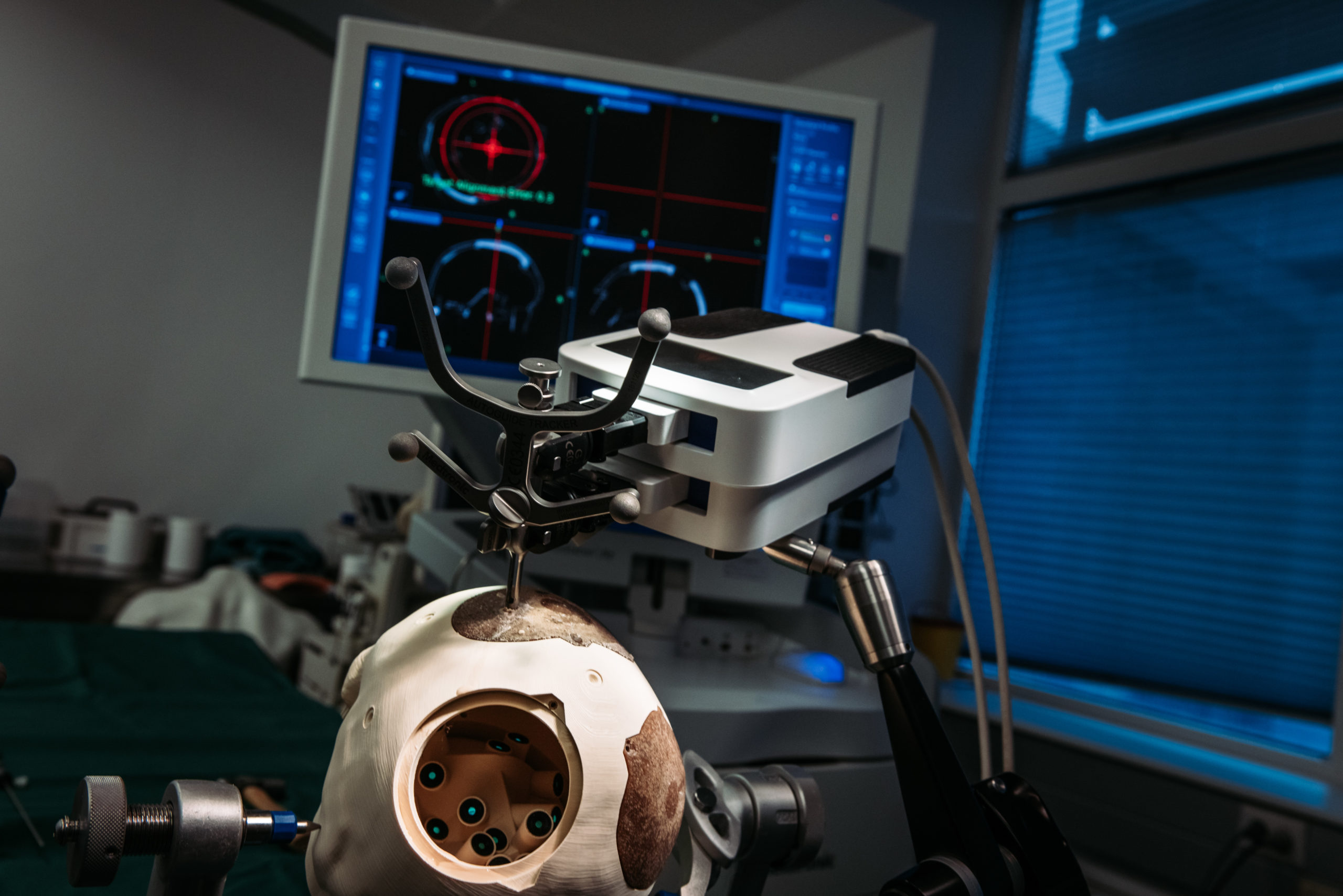
Therapy Planning and Workflow Support
This research area is dealing with new methods for planning of treatment in the pre-operative phase as well as with different aspects of workflow support in order to help the surgeon to sufficiently adhere to such a plan. In the deliberative control scheme of a therapy process, this area thus is covering the “planning” phase.
The topic “Therapy Workflow Support” is dealing with different aspects of workflow support, starting with an update of our current workflow assessment system, development of a generic workflow modelling toolbox, and implementation of a workflow engine. One core activity is generation of process specific process models and their use for workflow monitoring and workflow support systems. The aforementioned modules will be integrated to demonstrators in different configurations and for different clinical use cases.
The topic “Advanced Planning Concepts” basically deals with two different approaches for pre-operative planning. Investigation of software tools for planning in particular spotlights the integration of simulation methods into the planning procedure for immediate estimation of the effect of treatment and iterative improvement of the plan. In addition, the potential use of pre-existing data/knowledge for optimal planning will be investigated. Furthermore, different aspects and options of the use of Additive Manufacturing for pre-operative planning will be analyzed in detail – including the application of this planning method in selected use cases.
Your Contact Person

Tamás Haidegger
Prof. Dipl.-Ing. Dr.techn.
Therapy Planning and Workflow Support, Surgical Data Science
+43 2622 22859 0tamas.haidegger@acmit.at
FOSMON
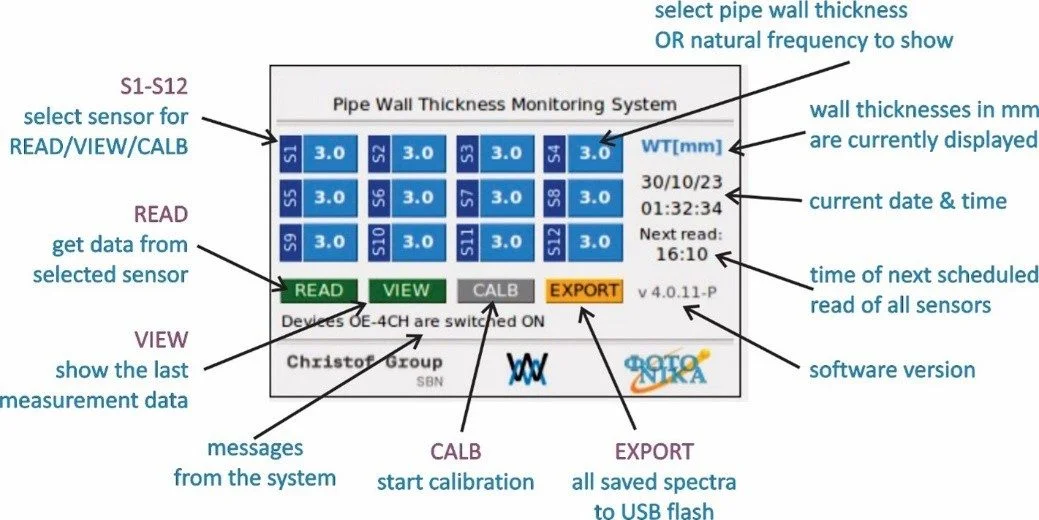
Test and evaluation of a fiber-optic sensor technology for in-situ and in-operando monitoring of heat exchanger tube wall thickness. (GFF project number: FTI21-A-005)
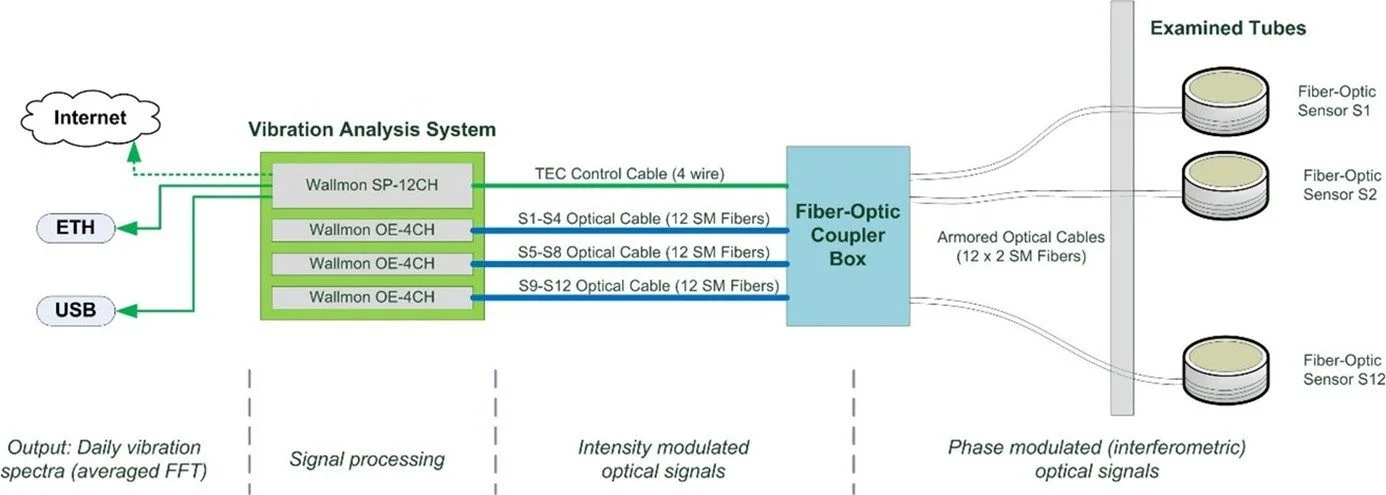
FOSMON measures in-situ and in-operando the wall thickness of heat-exchanging stainless steel tubes in harsh, high-pressure and high-temperature steam environments by applying FFT analysis to their vibration spectrum. The system uses fiber-optic interferometry, where a tightly wound single-mode fiber coil around the tube serves as an intrinsic stress and vibration sensor.
Project Start: November 2022
Project End: November 2025
Duration: 36 months
Funding: 282.945 €
Overall project costs: 435.300 €
Collaborating Partners:
Funding Body:
Gesellschaft für Forschungsförderung Niederösterreich m.b.H. (GFF NÖ)
Description:
- Research and development a method of non-destructive testing (NDT), structural health monitoring (SHM) and wall-thinning measurement of heat exchanger (HE) tubes in a stripper.
- This project proposes a new sensor system based on high-coherence interferometry, implemented in an “all-in-fibre” sensing configuration using single-mode optical fibres, for in-situ and in-operando monitoring of HE tubes wall thickness (see Figure below).
- Main hypothesis: a permanent loss of material mass induces a wall-thinning of the HE tubes that leads towards change of resonant frequency of the tube, shifting it toward higher values.

- The core idea of the proposed technique is to measure the resonant frequency of the tubes from the FFT power spectrum of interference signals obtained from fibre-optic sensors.
- The system has long-term applications in various industrial fields operating in harsh environments, such as corrosive atmospheres and high-pressure/high-temperature (HPHT) conditions.
- It contributes to increasing the safety of chemical plants by preventing structural failures caused by wall-thinning of tubes due to corrosion or erosion processes.
- To aim is to shift the current state-of-the-art paradigm from “Time-Based Maintenance” to “Condition-Based Maintenance”.
- The expected cost savings provide considerable potential benefits for end-users of such a wall-thinning monitoring system in similar markets.
References:
- Z. Djinović, A. Gavrilović-Wohlmuther, M. Tomić, A fiber-optic technique for the wall thickness measurement of the industrial tubes under harsh environment, 47th MIPRO ICT and Electronics Convention (MIPRO), Opatija, Cratia, 20-24 May 2024, DOI: 10.1109/MIPRO60963.2024.1056927
- M.Tomić, A. Gavrilović-Wohlmuther, Z.Djinović, Measurement of Wall Thickness in Heat Exchanger Tubes by Simultaneous Use of Low- and Highcoherence Fiber-Optic Interferometry, 48th MIPRO ICT and Electronics Convention (MIPRO), 02-06 June 2025, DOI: 10.1109/MIPRO65660.2025.11131780
- EP000004310434B1 HEAT EXCHANGER WITH FIBRE OPTIC SENSOR FOR DETERMINING A THICKNESS OF A HEAT EXCHANGER TUBE OF THE HEAT EXCHANGER AND METHOD FOR OPERATING SUCH A HEAT EXCHANGER
- WO002024017511A1 HEAT EXCHANGER COMPRISING A FIBRE-OPTIC SENSOR FOR DETERMINING A TUBE WALL THICKNESS OF A HEAT-TRANSFER TUBE OF THE HEAT EXCHANGER AND METHOD FOR OPERATING SUCH A HEAT EXCHANGER
- ASHMOSD-I, Austrian Structural Health Monitoring System Demonstrator; FFG-BMVIT, Takeoff Program, Project No: 814579
- ASHMOSD-II, Austrian Structural Health Monitoring System Demonstrator; FFG-BMVIT, Takeoff Program, Project No: 830397, Follow up
For more details, please see: FOSMON Brochure
The content does not necessarily represent the views of the State of Lower Austria or the Lower Austrian Research Promotion Agency as the funding agency. Therefore, neither the State of Lower Austria nor the funding agency can be held responsible for the content.
For more information:
Cornelia Häusler MSc.
Email: cornelia.haeusler@acmit.at
For business interests:
Dr. Aleksandra Gavrilovic-Wohlmuther
Email: a.gavrilovic-wohlmuther@christof-group.com
Mob: +43 664 80 883 4145
DEBRIDEMENT 2.0
A semi-automated robotic system challenges traditional assumptions
Appropriate wound debridement plays a key role in wound management and healing. In this study, we developed and evaluated a novel clinic-related in-vitro test system for detailed analysis of mechanical wound debridement procedures. The application-oriented in-vitro test system comprises a debridement process tracking device and a robotic debridement simulation device together with artificial wounds. Wound debridement movements, number of movement cycles, velocities as well as contact forces of medical experts were recorded in real-time by the debridement tracking device and reproduced with the robotic system. Our in-vitro test system has been evaluated through mechanical debridement product testing. Efficacy of wound debridement and product uptake were analyzed. Reproducibility and statistical significance of obtained in-vitro results verified the functionality and suitability of our system. Data obtained from our system can be used for optimization of wound debridement strategies by improving existing products and procedures or by developing novel approaches. In its entirety, our innovation challenges debridement analysis systems that exclude real-time individuality of debridement procedures. The semi-automated robotic system enables an effective learning process, from real-time tracking to in-vitro testing and finally back to real-time application. This new approach provides a strong link between in-vitro analysis and clinical applications.
Project Team: C. Häusler, F. Fussenegger, A. Hahnekamp, C. Kment, J. Schnur, A. Unger, H. Weißenböck
Transition to 3D Digital Pathology (3DPATH)
Partners:
- Clepio Biotech S.r.l., Italy
- University of Bern, Switzerland
ACMIT contribution:
- Technology industrialization and compliance with IVD regulations
- Regulatory, usability, risk management
- Manufacturing of small series of product-equivalent 3DPATH components in full conformance with ISO 13485
- Validation and demonstration of 3DPATH system
- Simulation for optical system design
- Assembly of optical systems, and measurement systems for quality assurance during and
after assembly
Description:
- 3D histopathology scanner for clinical tissue evaluation, improving reliability, throughput, and scalability
- Innovative Light-Sheet Fluorescence Microscopy (LSFM) and fast data analysis technology with autofocus, multi-wavelength imaging with high degree of novelty and performance
- Economic and societal impact of improved pathology on cancer-related diagnostic decisions
- Accurate diagnosis providing improved quality of patient care
Artificial Intelligence and Robotics for Prostate Biopsy (ROBIOPSY)
Partners:
- University of Verona, Italy
- Needleye Robotics S.r.l., Italy
- Department of Urology of the University of Freiburg – Medical Centre (UKLFR), Germany
- Day One – Funding & Venture Building for Technology Transfer projects, Italy
ACMIT contribution:
- User centered design
- Regulatory affairs management
- System specifications, architecture, integration & verification
- Usability evaluation
- Development of robot hardware & control system
- Development of planning & monitoring software
- Risk management
- Implementation of a QMS
Description:
- Development of a robotic device for prostate cancer biopsy as a diagnostic tool ready for clinical trials
- A multi-centric clinical study for automatic prostate segmentation and lesion identification
- Analyse of prostate biopsy’s business case in selected European countries to time/cost reduction
- Easy design for a shorter procedure and training of the medical personnel
- More precision, safety, repeatability thanks to AI
- Reduction of target identification and positioning errors
- Early diagnosis of prostate cancer to improve patients' quality of life and reduce mortality rates
Artificial Intelligence Guidance for Robot-Assisted Eye Surgery (GEYEDANCE)
Partners:
- University of Verona, Altair Robotics Laboratory, Italy
- PRECEYES BV, Netherlands
- University of Ferrara, Italy
- University of Bern, ARTORG Center for Biomedical Engineering Research, Switzerland
- ZEISS Corporate Research and Technology (CRT), Germany
- ZEISS Medical Technology (MED), Germany
ACMIT contribution:
- Coordination of the project
- Performation of translational R&D for new therapy options, including regulatory affairs management
- PRECEYES’s robotic system will be used and adapted to include a unique intraocular distance-sensing modality developed by ACMIT
- Development and evaluation of the Artificial Intelligence (AI) - guidance system during assessment of the clinical and usability requirements
- Formulation of the use scenarios
Description:
- GEYEDANCE AI - based system supports retinal surgeons to optimize their task, in order to reduce the mental and physical load of the surgeon
- Contribution of a better surgical outcome for the patients
- Generation of tangible and “close-to-certification” technology being evaluated in the framework of a first clinical trial.
- CE certified robot platform PRECEYES Surgical System
- A prototype for a Common-Path optical coherence tomography (OCT) system with extended measurement depth from ACMIT
- Development of AI-components
- A board of world-wide leading eye surgeons, led by University of Ferrara, provide user insight and evaluate the results
Mimicking Adaptation and Plasticity in WORMS (MAPWORMS)
Partners:
- Sant’Anna School of Advanced Studies, The BioRobotics Institute, Italy
- National Inter-University Consortium for Marine Sciences, Italy
- Hellenic Centre for Marine Research, Institute of Marine Biology, Biotechnology and Aquaculture, Greece
- Hebrew University of Jerusalem, Chemistry Department, Israel
- Vexlum Ltd., Finland
ACMIT contribution:
- Translational R&D for new therapy options
- Development of sensor systems for surgical procedures
- Development of a multi-material 3D printer
Description:
- Developing the first bioinspired shape-morphing robot based on stimuli-responsive materials
- Inspired by the marine Annelida
- Completely autonomous robot, able to fulfill different tasks by adapting to the environment
- Smart shape-memory hydrogels representing morphing robots combining with non-reactive elements
- US scanners with high-resolution transducers for realtime imaging of the actuator/robot elements
- Set of medical 3D tracking systems for realtime tracking of actuator/robot elements
- Different 3D printers for rapid prototyping
- Experimental OR for validation/demonstration of selected biomedical use-cases
- Tools and evaluation setup for optical and fiber-optical elements
Medical Device Obligations Taskforce (MDOT)
Partners:
- Fraunhofer Gesellschaft zur Förderung der Angewandten Forschung E.V., Germany
- DEMCON advanced mechatronics Zuid B.V., Netherlands
- Borm Nanoconsult Holding BV, Netherlands
- FUNDACION CIDETEC, Spain
- Manegold Christoph-Robert, Germany
- Medizinische Hochschule Hannover, Germany
- University of Southampton, UK
- University of Linz, Austria
- Uniwersytet Medyczny im Karola Marcinkowskiego W Poznaniu, Poland
- University of Leeds, UK
- Mathys AG Bettlach, Switzerland
- University of Luxembourg, Luxembourg
ACMIT contribution:
- Minimally invasive surgery
- Database development
- Consultation node
- Quality and risk management
- Prototyping for clinical testing
Description:
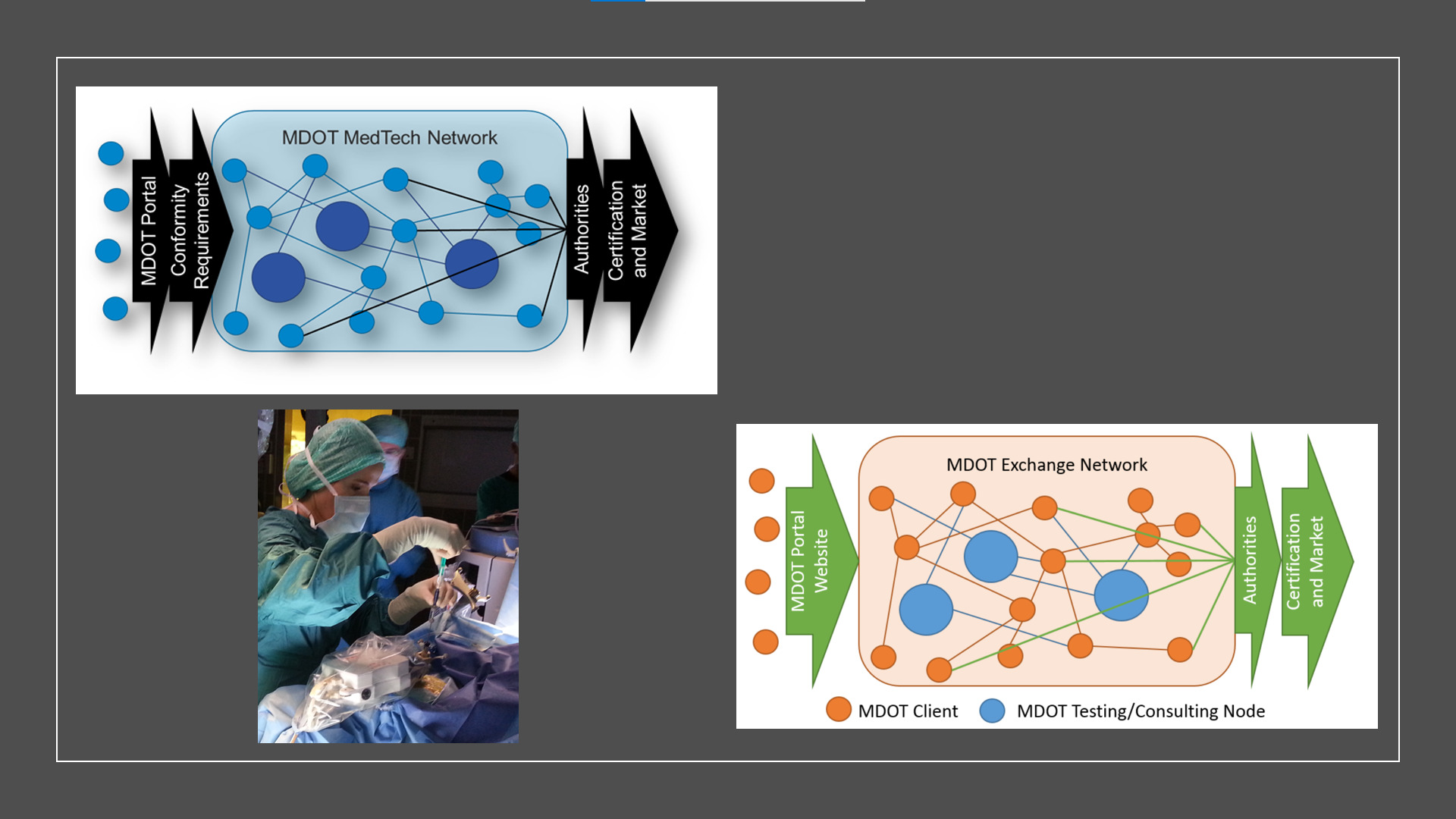
- Establishment of a data platform for simplified and affordable conformity and safety assessment tailored to the regulatory needs of MedTech Small and Mid-sized Enterprise (SMEs) in light of Medical Device Regulation (MDR)
- Improve and advance testing beds for medical devices in light of the MDR
- Reduce the burden of MDR for SMEs who cannot easily afford the required testing
- Make use of automation and prediction where possible and sensible
- The goal of MDOT is to establish a sustainable platform that enables MedTech SMEs:
- to establish a database of detailed testing data of commonly used components
- to safely and transparently exchange testing data using state-of-the-art technology to decrease cost while increasing quality and safety of medical device innovations
- to perform pre-clinical evaluations of commonly used parts together
- to get access to advanced testing and fabrication methods
Mathematical modeling of complex natural phenomena (DYNAMICS)
Partners:
- Politehnica University Timisoara, Romania
- Autonomous University of Barcelona, Spain
- Obuda University, Hungary
- EPAM Systems, Hungary
- Western University of Timisoara, Romania
- University of North Caroline at Charlotte, USA
- University of Sao Paulo at Sao Carlos, Brazil
- Shanghai Jiao Tong University, China
ACMIT contribution:
- Expertise related to potential applications of the project research in medical technologies
- Opportunities for secondments to project researchers to network
- Employment opportunities for the summer schools and other project participants
Description:
- Contributions to codimension k bifurcations in dynamical systems theory
- Development of new knowledge in the area of codim k bifurcations for continuous and discrete (smooth and non-smooth) dynamical systems
- Training opportunities in these areas for early-stage researchers
- Study of codim k bifurcations with ≥ 3 arising from degenerate Bautin bifurcations in continuous smooth dynamical systems
- Study codim k bifurcations with ≥ 3 arising from Hopf-Hopf (HH) bifurcations in continuous smooth dynamical systems
- Study codim k bifurcations in smooth dynamical systems arising from other bifurcations
- Study codim k bifurcations, ≥ 1, in non-smooth dynamical systems
- Study of discontinuous piecewise differential systems and continuous and discrete non-smooth dynamical systems resulting from modelling oscillators with impacts
Interactive Robotic Endoscope Guidance in Neurosurgery (ENDOROB)
Partners:
- University of Twente, Netherlands
- Charité – University Medicine Berlin, Department of Pediatric Neurosurgery, Germany
- Medical University of Vienna, Department of Neurosurgery, Austria
ACMIT contribution:
- Coordinator of the project
- Registered Digital Innovation Hubs in Healthcare Robotics (DIH Hero) Service Provider with DIH Services
- Development of a demonstrator for endoscope guidance
- Product certification support for EU and non-EU markets –analysis of applicable standards, integrated Risk and Usability Management, and ISO 13485-conform documentation
- Provider for own cloud data server
- Translation of the results to the applications in the field of neurosurgery, such as ENT surgery, spine surgery, or even to visceral surgery
Description:
- Interactive robotic guidance of a neurosurgical endoscope
- Demonstrator for a novel setup, based on an existing small and lightweight robot platform
- Validated final setup in a real clinical scenario
- Advantageous for workflow integration and accessibility to the surgical field
- Applications in endoscopically assisted neurosurgical procedures: endoscopic transsphenoidal approach for endosellar or parasellar pituitary surgery and the third ventriculostomy
Distance Measurement for Robot-Assisted Surgery (DIMRAS)
Partners:
- PRECEYES BV, Netherlands
ACMIT contribution:
- Supporting the development and certification of robot system for needle based interventions
- Development and OEM (Original Equipment Manufacturer) production of a robot for CT/CBCT guided procedures in interventional radiology
- Development partner for a novel robot system in neurosurgery, which received FDA clearance in November 2019 and was marketed by the world leader of neuronavigation systems
- Translate the DIMRAS results to RAS applications outside the field of ophthalmology, such as brain tumor surgery
Description:
- ERM (epiretinal membrane) can be treated by peeling the epiretinal membrane and vitrectomy
- The instrument developed in DIMRAS will enable the robot to define forbidden regions, to prevent retinal touches during vitrectomy, and to impose an upper limit on the retinal indentation during membrane peeling
- Transfer of the OCT (optical coherence tomography) technology from the field of diagnostics into a specific clinical sensor
- Instrument-integrated distance sensor for robot-assisted tool manipulation in vitreoretinal surgery
- Sensor-based robotic method for improvement of speed and precision (semi) automated navigation by the instrument during an eye operation
- Increase of the reproducibility and safety for membrane peeling and vitrectomy
Smart Autonomous Robotic Assistant Surgeon (SARAS)
Partners:
- University of Verona (UNIVR), Italy
- University of Moderna and Reggio Emilia (UNIMORE), Italy
- University of Ferrara (UNIFE), Italy
- Ospedale San Raffaele (OSR), Italy
- Universitat Politecnica de Catalunya (UPC), Spain
- University of Dundee (UNIVDUN), UK
- Oxford Brookes University (OBU), UK
- Medineering GmbH (MEDIN), Germany
ACMIT contribution:
- System integration and phantom augmentation
- Development of multi-functional tools, medical robotics, training systems for medical/surgical education and training, as well as workflow analysis
- Technical specification and optimization
- Usability engineering and support of company partners with product certification
- Support of dissemination activities with its network of clinical partners and hospital operators
- Open new markets in surgical robotics, phantoms design and training to commercially disseminate the project results
- Certified development processes (ISO 13485)
Description:
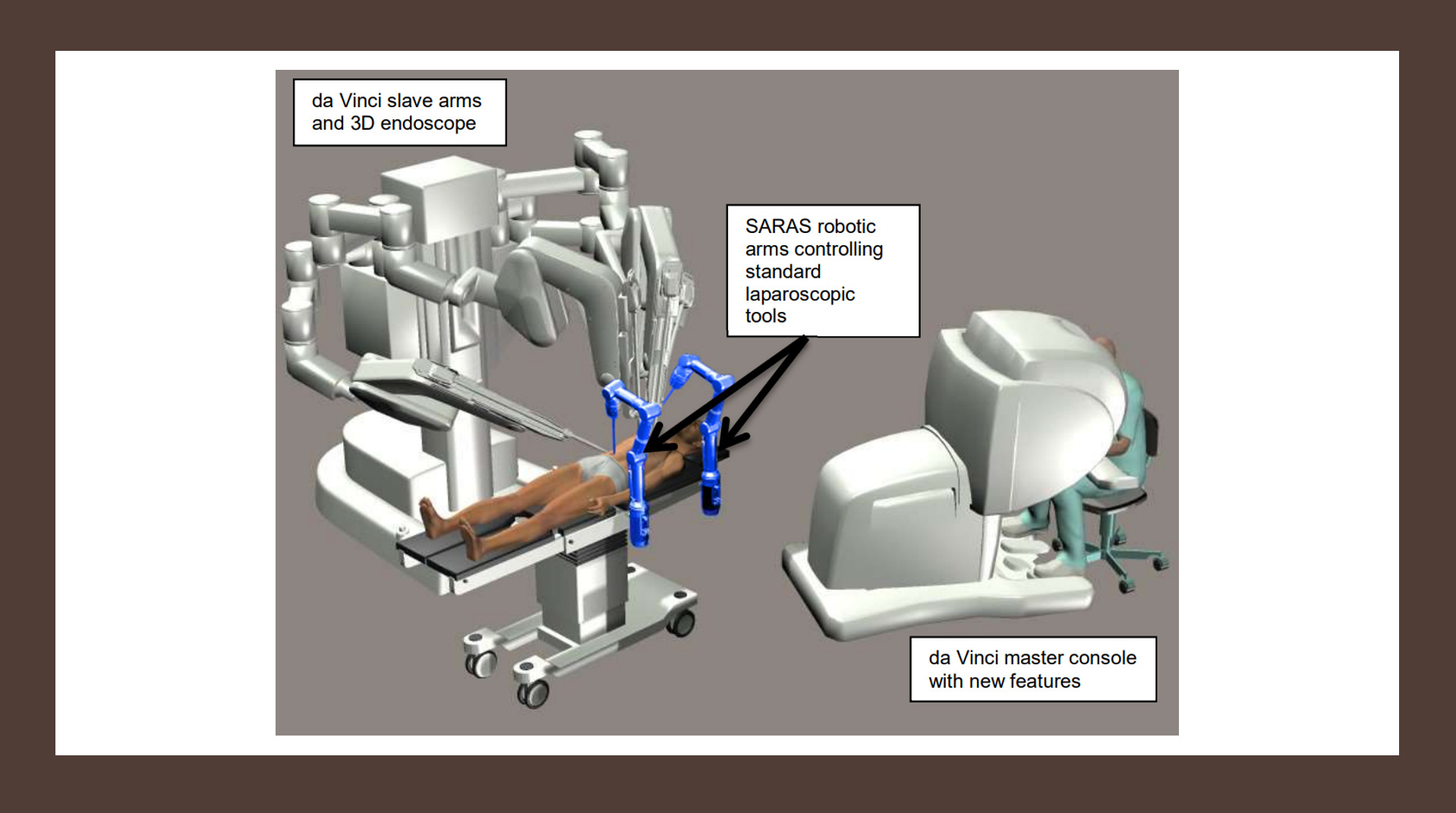
- Development of the next-generation of surgical robotic systems with a single surgeon to execute Robotic Minimally Invasive Surgery (R-MIS) without the need of an expert assistant surgeon
- Solo-surgery system consisting of a pair of cooperating and autonomous robotic arms holding the surgical instruments
- Leverage on a ground-breaking artificial intelligence (AI) module to develop a cognitive robotic system capable of autonomously understanding the present and future surgical situation, and performing its predefined actions at the right place and time
- SARAS assistive robotic arms are able to perform the same tasks that are now carried out by the assistant surgeon during a robotic or a laparoscopic procedure
Artificial Intelligence-driven diagnosis and treatment of Upper Aerodigestive Tract Cancers (AIRCARE)
Partners:
- Italian Institute of Technology, Italy
- KU Leuven, Belgium
- University of Southern Denmark, Denmark
- Sant’Anna, School of Advanced Studies – Pisa, Italy
- University of Genoa, San Martino Hospital, Italy
- IDIBAPS, Hospital Clinic of Barcelona, Spain
- El.En. Group, Italy
- BARCO NV, Belgium
- Inventya Ventures, Ireland
- TimeLex, Belgium
- D'Annunzio University, Italy
ACMIT contribution:
- Integration of individual input elements into a central surgeon console
- Regulatory compliance
- Documentation of the certification process
Description:
- Development of AI-driven diagnosis and treatment of Upper Aerodigestive Tract Cancers (UADT)
- Introduction of advanced AI-driven and robotic technologies into clinical workflows
- AI-powered Integrated Diagnostic System for real-time detection and diagnosis
- Early detection and in situ cancer characterization
- Integrated Therapy System for enhanced recognition of tumour margins and increased surgical precision
- AI-assisted tumour segmentation, robotic laser steering and advanced robotic forceps controlled by an ergonomic console greater safety and accuracy